Scientists at Albert Einstein College of Medicine
Newly Developed Fluorescent Protein Makes Internal Organs Visible
July 17, 2011 – (BRONX, NY) – Researchers at Albert Einstein College of Medicine of Yeshiva University have developed the first fluorescent protein that enables scientists to clearly "see" the internal organs of living animals without the need for a scalpel or imaging techniques that can have side effects or increase radiation exposure.

Vladislav Verkhusha, Ph.D. (left) and Grigory Filonov, Ph.D. (right)The new probe could prove to be a breakthrough in whole-body imaging – allowing doctors, for example, to noninvasively monitor the growth of tumors in order to assess the effectiveness of anti-cancer therapies. In contrast to other body-scanning techniques, fluorescent-protein imaging does not involve radiation exposure or require the use of contrast agents. The findings are described in the July 17 online edition of Nature Biotechnology.
For the past 20 years, scientists have used a variety of colored fluorescent proteins, derived from jellyfish and corals, to visualize cells and their organelles and molecules. But using fluorescent probes to peer inside live mammals has posed a major challenge. The reason: hemoglobin in an animal''s blood effectively absorbs the blue, green, red and other wavelengths used to stimulate standard fluorescent proteins along with any wavelengths emitted by the proteins when they do light up.
To overcome that roadblock, the laboratory of Vladislav Verkhusha, Ph.D., associate professor of anatomy and structural biology at Einstein and the study''s senior author, engineered a fluorescent protein from a bacterial phytochrome (the pigment that a species of bacteria uses to detect light). This new phytochrome-based fluorescent protein, dubbed iRFP, both absorbs and emits light in the near-infrared portion of the electromagnetic spectrum – the spectral region in which mammalian tissues are nearly transparent.
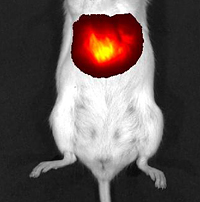
Liver cells in this mouse contain the fluorescent protein iRFP. The mouse was exposed to near-infrared light, which has caused iRFP to emit light waves that are also near-infrared. The composite image shows these fluorescent near-infrared waves passing readily through the animal’s tissues to reveal its brightly glowing liver. The researchers targeted their fluorescent protein to the liver – an organ particularly difficult to visualize because of its high blood content. Adenovirus particles containing the gene for iRFP were injected into mice. Once the viruses and their gene cargoes infected liver cells, the infected cells expressed the gene and produced iRFP protein. The mice were then exposed to near-infrared light and it was possible to visualize the resulting emitted fluorescent light using a whole-body imaging device. Fluorescence of the liver in the infected mice was first detected the second day after infection and reached a peak at day five. (See accompanying image.) Additional experiments showed that the iRFP fluorescent protein was nontoxic.
"Our study found that iRFP was far superior to the other fluorescent proteins that reportedly help in visualizing the livers of live animals," said Grigory Filonov, Ph.D., a postdoctoral fellow in Dr. Verkhusha''s laboratory at Einstein, and the first author of the Nature Biotechnology paper. "iRFP not only produced a far brighter image, with higher contrast than the other fluorescent proteins, but was also very stable over time. We believe it will significantly broaden the potential uses for noninvasive whole-body imaging."
Dr. Filonov noted that fluorescent-protein imaging involves no radiation risk, which can occur with standard x-rays and computed tomography (CT) scanning. And unlike magnetic resonance imaging (MRI), in which contrasting agents must sometimes be swallowed or injected to make internal body structures more visible, the contrast provided by iRFP is so vibrant that contrasting agents are not needed.
The study, "Bright and stable near-infrared fluorescent protein for in vivo imaging," was published in the July 17 online edition of Nature Biotechnology. Other Einstein researchers involved in the study were Kiryl Piatkevich, Ph.D., Li-Min Ting, Ph.D., Jinghang Zhang, M.D., and Kami Kim, M.D. This research was carried out at the Gruss Lipper Biophotonics Center and supported by grants from the National Institute of General Medical Sciences of the National Institutes of Health.
Other Top Stories
9/11 World Trade Center Exposure Linked to Heart Disease Among NYC Firefighters
On Becoming a Physician: New Einstein Students Receive White Coats and Stethoscopes
Novel Therapy for Acute Migraine Shows Promise in Phase 3 Clinical Trial
First Complete Wiring Diagram of an Animal's Nervous System
Multimillion Dollar NIH Grant to Help Reduce Opioid Use & Get Care to People Who Need It
NIH Grant Funds $23 Million Study of Diseases Affecting People Living with HIV
New TAILORx Data Guides Adjuvant Therapy in Younger Breast Cancer Patients
Einstein Celebrates Its 61st Commencement
Bolstering Biopsies: Testing Patients' Individual Cells to Guide Treatment



Tablet Blog