Nanoparticle Wound Therapy
Nanoparticle Therapy Promotes Wound Healing
March 26, 2015—(Bronx, NY)—An experimental therapy developed by researchers at Albert Einstein College of Medicine of Yeshiva University cut in half the time it takes to heal wounds compared to no treatment at all. Details of the therapy, which was successfully tested in mice, were published online in the Journal of Investigative Dermatology.
"We envision that our nanoparticle therapy could be used to speed the healing of all sorts of wounds, including everyday cuts and burns, surgical incisions, and chronic skin ulcers, which are a particular problem in the elderly and people with diabetes," said study co-leader David J. Sharp, Ph.D., professor of physiology & biophysics at Einstein.
Dr. Sharp and his colleagues had earlier discovered that an enzyme called fidgetin-like 2 (FL2) puts the brakes on skin cells as they migrate towards wounds to heal them. They reasoned that the healing cells could reach their destination faster if their levels of FL2 could be reduced. So they developed a drug that inactivates the gene that makes FL2 and then put the drug in tiny gel capsules called nanoparticles and applied the nanoparticles to wounds on mice. The treated wounds healed much faster than untreated wounds.

David Sharp, Ph.D.FL2 belongs to the fidgetin family of enzymes, which play varying roles in cellular development and function. To learn more about FL2's role in humans, Dr. Sharp suppressed FL2's activity in human cells in tissue culture. When those cells were placed on a standard wound assay (for measuring properties like cell migration and proliferation), they moved unusually fast. "This suggested that if we could find a way to target FL2 in humans, we might have a new way to promote wound healing," said Dr. Sharp.
Dr. Sharp and project co-leader Joshua Nosanchuk, M.D., professor of medicine at Einstein and attending physician, infectious diseases at Montefiore Medical Center, developed a wound-healing therapy that uses molecules of silencing RNA (siRNAs) specific for FL2. The siRNAs act to silence genes. They do so by binding to a gene's messenger RNA (mRNA), preventing the mRNA from being translated into proteins (in this case, the enzyme FL2). However, "siRNAs on their own won't be effectively taken up by cells, particularly inside a living organism" said Dr. Sharp. "They will be quickly degraded unless they are put into some kind of delivery vehicle."
To find a way to deliver siRNAs for curbing FL2, Dr. Sharp collaborated with Joel Friedman, M.D., Ph.D., professor of physiology & biophysics and of medicine at Einstein, and study co-leader Adam Friedman, M.D., director of dermatologic research at Einstein and Montefiore, who together had developed nanoparticles that protect molecules such as siRNA from being degraded as they ferry the molecules to their intended targets.
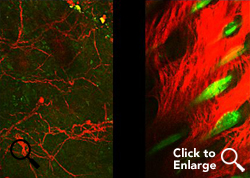
Imaging of burns indicates that those treated with the FL2 inhibitor nanotechnology experienced collagen deposition and hair follicle formation. (2-photo confocal microscopy)
Credit: Vera DesMarais/Albert Einstein College of MedicineThe nanoparticles with their siRNA cargoes were then tested by topically applying them to mice with either skin excisions or burns. In both cases, the wounds closed more than twice as fast as in untreated controls. "Not only did the cells move into the wounds faster, but they knew what to do when they got there," said Dr. Sharp. "We saw normal, well-orchestrated regeneration of tissue, including hair follicles and the skin's supportive collagen network."
Dr. Sharp plans to start testing the therapy on pigs, whose skin closely resembles that of humans, within months.
The paper, which published online March 10, is titled "Fidgetin-like 2: a novel microtubule-based regulator of wound healing." The other contributors, all at Einstein, are: Rabab Charafeddine, Ph.D. candidate; Joy Makdisi, M.D. student; David Schairer, M.D.; Brian O'Rourke, Ph.D..; Juan D. Diaz-Valencia, Ph.D.; Jason Chouake, M.D. student; Allison Kutner, M.D.; Aimee Krausz, M.D. student; Brandon Adler, M.D. student; Parimala Nacharaju, Ph.D.; Hongying Liang; Suranjana Mukherjee, Ph.D. candidate; and Joshua Nosanchuk, M.D.
The research presented in this manuscript was funded by grants from the National Institute of Health (R01 GM109909) and the Telemedicine and Advanced Technology Research Center (TATRC) at the U.S. Army Medical Research and Materiel Command (USAMRMC) through award W81XWH1210379. Support was also granted by the Women's Dermatologic Society academic grant program.
David Sharp and Rabab Charafeddine are co-inventors on U.S. Patent #20130022667, "Fidgetin-like 2 as a target to enhance wound healing," which has been licensed by MicroCures Inc. Dr. Sharp is currently chief scientific officer of MicroCures. Dr. Sharp, Dr. Joel Friedman, Dr. Adam Friedman and Dr. Nosanchuk all have equity in the company. The other authors declare no conflicts of interest.
Other Top Stories
9/11 World Trade Center Exposure Linked to Heart Disease Among NYC Firefighters
On Becoming a Physician: New Einstein Students Receive White Coats and Stethoscopes
Novel Therapy for Acute Migraine Shows Promise in Phase 3 Clinical Trial
First Complete Wiring Diagram of an Animal's Nervous System
Multimillion Dollar NIH Grant to Help Reduce Opioid Use & Get Care to People Who Need It
NIH Grant Funds $23 Million Study of Diseases Affecting People Living with HIV
New TAILORx Data Guides Adjuvant Therapy in Younger Breast Cancer Patients
Einstein Celebrates Its 61st Commencement
Bolstering Biopsies: Testing Patients' Individual Cells to Guide Treatment



Tablet Blog