

Global Health
Malaria: Progress and Promise
Einstein-Montefiore researchers seek new treatments, with a special focus on cerebral malaria
As old infectious diseases like TB spread and new ones like SARS arise, sometimes it seems we’re losing the fight against disease-causing microbes. But there’s heartening news on at least one front: malaria. Since 2000, the infection rate from this mosquito-borne illness has plummeted by 37 percent and the death rate by 60 percent, according to the World Health Organization.

Kami Kim, M.D.These dramatic reductions can be traced to several interventions, including the distribution of bed nets, mosquito control, aggressively tracking down cases of malaria, and, perhaps most important, the development of a potent anti-malarial drug called artemisinin (which earned its discoverer a share of the 2015 Nobel Prize in Physiology or Medicine).
“This is a great example of what can be accomplished when governments, public health organizations, private philanthropists, doctors and scientists all work together toward a common goal,” said Kami Kim, M.D., a longtime malaria researcher and professor of medicine, of microbiology & immunology, and of pathology at Einstein and attending physician, infectious disease at Montefiore.

Johanna Daily, M.D., M.S.But this favorable trend may not continue. Resistance to artemisinin—now the cornerstone anti-malarial therapy—is emerging in Southeast Asia. “There’s no resistance yet in Africa, but there will be,” said malaria expert Johanna Daily, M.D., M.S., associate professor of medicine and of microbiology & immunology at Einstein and attending physician, infectious disease at Montefiore. “This could be a huge problem because there’s no next anti-malarial ready to replace artemisinin.”
Meanwhile—despite reductions in incidence and deaths—malaria remains a significant threat to public health. Some 200 million people are infected each year, causing about 438,000 deaths, largely in sub-Saharan Africa. The vast majority of those deaths occur in children under age five, primarily because of a complication called cerebral malaria, or CM.
Of the five species of the malaria parasite that can infect humans, only Plasmodium falciparum—the most deadly malaria species—causes this complication. While the mechanisms by which CM injures the brain are not well understood, problems begin when red blood cells infected with P. falciparum sequester within the brain’s microvasculature--adhering to the endothelial lining, to each other and to healthy red blood cells as well. The sequestered mass of red cells impairs blood flow and oxygen delivery to brain tissue, setting the stage for seizures and coma.

A mother and her comatose child with cerebral malaria on the Malaria Research Ward at Queen Elizabeth Central Hospital, Blantyre, Malawi. Photo credit: Jim Peck, Michigan State University.At least 575,000 children in Africa develop cerebral malaria each year, and about one in eight of them die. Up to a quarter of the survivors experience long-term neurological and cognitive problems, including memory loss, learning and language difficulties, movement deficits and psychiatric disorders.
CM is highly treatable if diagnosed and treated early—with an emphasis on if. CM is difficult to diagnose, and few patients in resource-poor areas like sub-Saharan Africa get timely treatment—some none at all.
Malaria and Monocytes
In light of this ongoing tragedy, a number of researchers around Einstein have devoted their attention to CM. Dr. Kim, for example, is trying to understand why some kids succumb to CM and others do not. Inflammation is thought to be a contributing factor, and so is HIV, but with few patients available for study, definitive answers have been hard to come by.
More clues are beginning to emerge, thanks in part to the Blantyre Malaria Project in Malawi, which for decades has followed thousands of children with CM, including many co-infected with HIV. Through the project, Dr. Kim was able to obtain and analyze autopsied brain tissue collected from more than 100 children who had succumbed to coma caused by CM or other conditions.
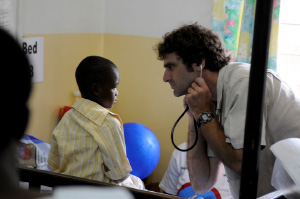
Dr. Karl Seydel (MSU and Blantyre Malaria Project) about to examine a young patient recovering from cerebral malaria on the Malaria Research Ward at Queen Elizabeth Central Hospital, Blantyre, Malawi. Photo credit: Jim Peck, Michigan State University.Dr. Kim and her colleagues found that coma patients with CM had more than nine times as many monocytes (cells associated with inflammation) and platelets (cells that promote blood clotting) in their blood than did coma patients who did not have malaria. Among those children who had CM, HIV-positive children had twice the number of monocytes and platelets in their blood compared with HIV-negative children.
“Our study doesn’t prove that monocytes and platelets cause CM,” said Dr. Kim, whose findings were reported in the journal mBio and covered in the health section of the New York Times. “But the fact that these cells were present in huge amounts when there were large numbers of parasites strongly suggests that they’re contributing to the condition. We never see this particular accumulation of cells in healthy brain tissue.”
The findings suggest that treatments to reduce inflammation or clotting, or both, may help patients with CM or with CM-HIV co-infection. In addition, HIV-positive children at risk for malaria may benefit from targeted malaria-prevention efforts, according to the study’s lead author Sarah Hochman, M.D., adjunct clinical assistant professor of medicine at Einstein.
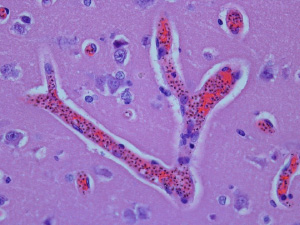
Brain section from an autopsy on a person with cerebral malaria shows small blood vessels clogged with infected red blood cells. Malaria pigment within malaria parasites is visible as small dark dots inside infected red cells. Mononuclear cells (dyed purple) are also inside the vessels. Photo credit: Sarah Hochman, M.D., Einstein.In future studies, Dr. Kim’s team hopes to identify specific signatures—e.g., ratios of monocytes, platelets, and other blood factors—that could be used for diagnosing CM and for predicting which patients are particularly susceptible to fatal complications and thus may warrant more aggressive intervention.
Of Genomes and Metabolomes
Dr. Kim’s research dovetails with that of Dr. Daily, who is studying why only a small percentage of malaria patients with parasite-infected red blood cells go on to develop CM. Dr. Daily has hypothesized that CM occurs in those patients who mount an overzealous immune response to infection. But if so, what is driving the immune response?
Several years ago, Dr. Daily began to suspect that neutrophils, a type of white blood cell involved in mediating inflammation, may be responsible. But finding answers took a bit of doing. Neutrophils live for just 12 hours outside of the body and can’t be frozen. That meant that someone would have to go to the source—actual CM patients—to conduct a proper analysis. Dr. Daily had been to Africa many times to work in collaboration with the Blantyre Malaria Project (BMP). But this particular assignment fell to one of her doctoral students, Catherine M. Feintuch, who had a background in public health and a desire for first-hand international health experience.

Catherine M. Feintuch
“We’d take a blood sample from the patient with CM upon admission so it could be studied in detail and correlated with the patient’s outcome—and it didn’t matter what time of day or night,” said Dr. Feintuch, who spent 10 weeks at the BMP working in the laboratory and collecting samples, each of which required four hours of processing. “Our analysis suggested that neutrophils play a role in the host’s response to infection and may mediate disease outcomes,” she said.
Delving deeper into the host response, Dr. Feintuch (she recently received her Ph.D.) and her mentor analyzed gene expression in the blood of children with CM. Children with severe CM (comatose and with infected red cell sequestration in their brain microvasculature) were compared to children with milder CM (comatose but no sequestration of infected red cells and a lower death rate).
Only the group with severe CM exhibited a gene-expression profile consistent with a high inflammatory state, including higher neutrophil activation. In addition to having a low inflammatory state, most of the children with mild CM had unusually high levels of fetal hemoglobin (the form of hemoglobin that usually disappears from the circulation after six months of age and has been associated with protection from severe malaria).
Based on their data, the researchers hypothesize that activated neutrophils release damaging reactive oxygen species (also known as free radicals), among other substances. These substances, in turn, alter cell-surface receptors on platelets (blood cells involved in coagulation) and endothelial cells lining the microvasculature so that infected red blood cells stick to them, causing the brain’s microvasculature to become clogged and leading to impaired blood flow and brain swelling. (Dr. Feintuch’s paper on activated neutrophils and CM was just accepted by the journal mBio.)
“We have more to learn about the mechanisms by which parasite-infected red cells sequester in the brain microvasculature,” said Dr. Daily. “But our findings suggest that making endothelial cells and platelets less sticky might inhibit infected red cells from sequestering in the brain’s microvasculature and thereby lessen the damaging and lethal effects of CM. We have some ideas about how to do this and are testing molecules that may be protective.”
Dr. Daily’s lab is also investigating why some children with CM develop brain swelling—a critical question in light of recent research showing that brain swelling and the resulting increase in intracranial pressure is what ultimately kills these young patients.

Vasiliki Pappa, M.P.H.Examining the metabolomes (the full complement of metabolites) of the peripheral blood of children with CM, the researchers found that fatty acids called lysophospholipids are associated with brain swelling. The study also found a link between brain swelling and three inflammatory cytokines (proteins secreted by cells to carry signals to neighboring cells). These fatty acids and cytokines are known to alter endothelial cells and consequently increase the permeability of the blood-brain barrier. The results of this research were recently published in Malaria Journal (http://www.malariajournal.com/content/14/1/513).
“Our next step to make a blood-brain barrier model and screen for compounds that reduce its permeability,” said Vasiliki Pappa, M.P.H., a doctoral candidate in Dr. Daily’s lab, who did much of the research. “If we could find a way to inhibit or reduce the brain swelling in children with CM, that would be a major victory.”
Mice and Malaria
Mahalia S. Desruisseaux, M.D., assistant professor of pathology and of medicine at Einstein and attending physician, infectious disease at Montefiore, is investigating why many children with CM—even after successful treatment to eradicate the malaria parasite—experience neurological and cognitive deficits later in life.

Mahalia S. Desruisseaux, M.D.
Since it’s difficult to study certain aspects of the disease in humans, Dr. Desruisseaux has worked for the better part of a decade with a mouse model of CM. Using magnetic resonance scanning to image the brains of these mice, she has found that CM reduces cerebral blood flow, increases inflammation and the infiltration of white blood cells, further disrupts the blood-brain barrier and induces neuronal damage (specifically to axons, the portions of nerves that transmit impulses to other nerve cells). Her studies suggest that these effects are mediated by the protein endothelin-1 (ET-1), a key part of the ET-1 pathway involved in blood-brain barrier permeability, inflammation and vascular tone.
These findings indicate that therapies targeting components of the ET-1 pathway may help improve the neurological deficits associated with CM. In a 2012 study published in Life Sciences and involving mice with CM, Dr. Desruisseaux and her team tested the effects of HJP-272, an experimental endothelial cell-surface receptor blocker that they developed. When given in conjunction with artemether (a derivative of artemisinin), HJP-272 minimized changes in the brain’s microvasculature associated with CM’s neurological effects and improved survival.

Mahalia Desruisseaux, M.D. (r) with graduate student Brandi Freeman (l) and Tamiwe Tomoka.Former postdoctoral fellow Yuri C. Martins, M.D., Ph.D., along with Brandi D. Freeman, Ph.D. (a former graduate student in Dr. Desruisseaux’s lab), provided further evidence that the neurological and vascular disturbances seen in CM are at least partly due to ET-1 signaling. When mice infected with P. berghei—a strain of malaria that generally does not produce cerebral complications—were given the ET-1 protein, the animals developed the characteristic signs of CM. Treatment with an ET-1 receptor blocker prevented these effects. Dr. Freeman also showed that ET-1 activates the JNK pathway, known to play a role in neuronal death. Dr. Freeman presented her findings at the 2015 American Society of Tropical Medicine and Hygiene (ASTMH) Annual Meeting, where she was given the society’s Young Investigator Award.
In a different line of inquiry, Dr. Desruisseaux and former graduate student Henry Shikani have provided the first evidence that tau (a protein involved in the formation of damaging neurofibrillary tangles in Alzheimer's disease) plays a role in CM. The researchers found elevated levels of two forms of abnormal tau in CM mice. When the mice were given a drug known to clear these forms of tau in mouse models of Alzheimer’s, the drug prevented axonal damage in certain brain regions. The findings suggest that tau may be a good target for ameliorating neuronal damage and the subsequent neurocognitive impairment associated with CM.
Another former postdoctoral fellow in the Desruisseaux lab, Fernando P. Bruno, M.D., devised a novel imaging technique for assessing brain pathology in CM mice. Near-infrared fluorescent imaging allows researchers to noninvasively visualize neuroinflammation and blood-brain barrier disruption during the course of the disease. The technique may prove valuable in evaluating experimental therapies and in monitoring treatment. For his efforts, Dr. Bruno was awarded the 2013 ASTMH Young Investigator Award and was recognized by the Bill and Melinda Gates Foundation as a member of the “next generation” of promising scientists.
Going After Malaria’s Achilles’ Heel

Vern Schramm, Ph.D.Vern Schramm, Ph.D., and Myles Akabas, M.D., Ph.D., are developing drugs to kill the deadly malaria species P. falciparum. Both are targeting what is arguably the Achilles’ heel of malaria parasites: their inability to synthesize the purines that are vital for energy and for making DNA.
Since the parasites can’t synthesize those purines on their own, they need to import purine intermediates from the red blood cells they’ve invaded. Then they must convert those purine intermediates via enzymatic reactions into the purine bases they need: adenine and guanine for making DNA, as well as ATP and GTP for energy and metabolism.
Dr. Schramm’s approach is to short-circuit the pathway by which P. falciparum synthesizes purines from purine intermediates. He has developed a drug called BCX4945 to inhibit a vital component of that pathway: an enzyme called purine nucleoside phosphorylase (PNP).
BCX4945 belongs to a class of drugs known as transition state analogs, which Dr. Schramm, a professor of biochemistry at Einstein, has been developing since 1994. A transition state is the short-lived form of a chemical that occurs during a chemical change, for example, when an enzyme converts one chemical (the substrate) into another (the product).
After elucidating the transition-state structure for a particular enzyme, Dr. Schramm is able to design transition-state analogs to knock that enzyme out of action. The analogs closely resemble the actual transition-state structure but with one big difference: they powerfully inhibit the enzyme by binding to it and not letting go. The transition-state analog BCX4945 proved to have a high affinity for PNP.
After BCX4945 showed potency against laboratory cultures of P. falciparum, Dr. Schramm and colleagues tested the drug in three owl monkeys infected with a strain of P. falciparum that is consistently lethal without antimalarial therapy. Treatment with BCX4945 cleared the infections from all three animals within a week, and they remained parasite-free for up to nine days after treatment. The infection returned in the three monkeys after treatment ended, although with a lower rate of parasitic growth. No signs of toxicity were observed during the study period.
“Inhibiting PNP differs from all other current approaches for treating malaria,” said Dr. Schramm, whose results were reported in PLoS ONE. “For that reason, BCX4945 fits well with the current World Health Organization protocols for malaria treatment, which call for using combination-therapy approaches against the disease.”
Dr. Schramm has since worked with Dr. Daily and others at Einstein to understand the parasite’s response to BCX4945 and whether there is potential for resistance if the drug is used as a stand-alone therapy. Clinical testing of BCX4945 is still many years away.
Screening for New Anti-Malarials

Myles Akabas, M.D., Ph.D.Dr. Akabas, like Dr. Schramm, is also capitalizing on P. falciparum’s inability to synthesize its own purines. His strategy is to starve the parasite of the purine intermediates it needs to survive. His target: a parasite protein called PfENT1 that transports purine intermediates from blood cells into the parasites.
Drugs that block PfENT1 could conceivably kill the parasites. But an experimental approach for identifying PfENT1 inhibitors didn’t exist, until now.
Dr. Akabas is professor of physiology & biophysics, of medicine, and in the Dominick P. Purpura Department of Neuroscience at Einstein. He and his team—including two M.D.-Ph.D. students in his lab (I.J. Frame and Roman Deniskin) and colleagues at Einstein (Ian Willis, Ph.D., and Robyn Moir, Ph.D.) and Columbia University (Donald Landry, M.D., Ph.D., and David Fidock, Ph.D.)—developed a novel yeast-based high-throughput assay for identifying inhibitors of the PfENT1 transporter.
University of Malawi College of Medicine’s ICEMR Molecular and Genomics Core LaboratoryUsing the new technique, the researchers screened 64,560 different compounds, identifying 171 potential antimalarial drugs. Studies of nine of the most potent drugs showed that they kill P. falciparum parasites in laboratory culture.
“We’ve shown that the PfENT1 transporter is a potential drug target for developing novel antimalarial drugs,” said Dr. Akabas, whose findings were published earlier this year in ACS Chemical Biology. “By using our rather simple approach, scientists could create similar high-throughput screens to identify inhibitors for killing other parasites that rely on transporters to import essential nutrients.”
In January 2015, the National Institutes of Health awarded Dr. Akabas and his Columbia collaborators a five-year, $3.45-million grant to use his high-throughput assay to find and develop antimalarial drugs. Einstein has applied for patents to cover this assay.
Looking Ahead
The malaria parasite has proven a formidable foe, able to outwit almost every preventive or therapeutic thrown its way. Nevertheless, scientists are optimistic about ultimately gaining the upper hand.
“If philanthropic organizations like the Bill and Melinda Gates Foundation stay in the game, there’s a good chance we can decrease malaria transmission even further,” said Dr. Daily. “The number of cases should also decline as Africa becomes wealthier and housing improves, simply because living in a house with screens means less exposure to disease-carrying mosquitoes.” And, perhaps, malaria researchers will come up with the next big drug, vanquishing or at least controlling this ancient plague.
Drs. Kim, Daily and Desruisseaux all received grants from Einstein’s Global Health Center to support their research with the Blantyre Malaria Project in Malawi.
Posted on: Wednesday, January 20, 2016

Tablet Blog